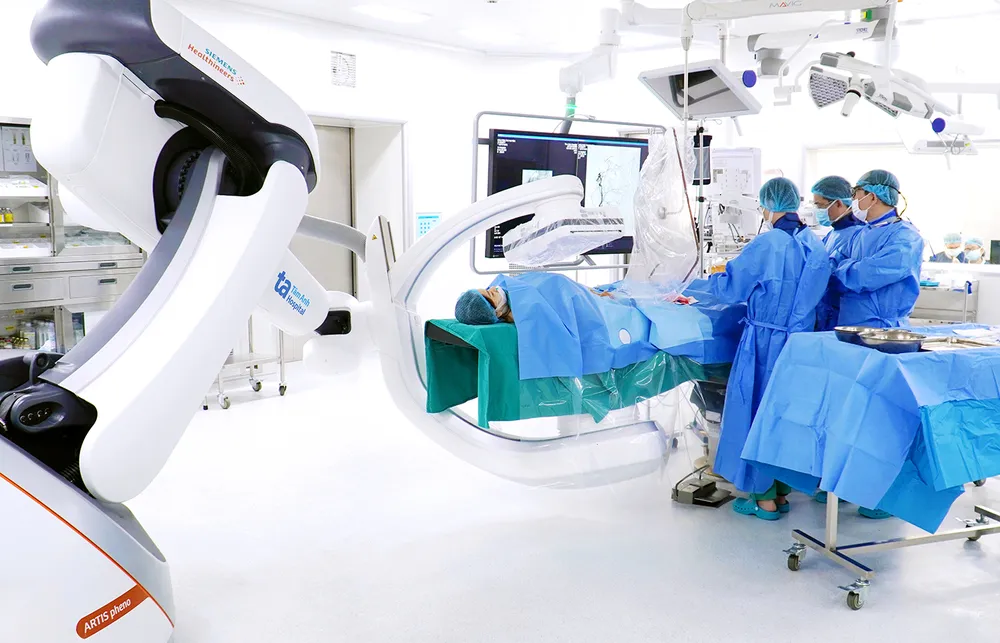
Lower costs, more treatment opportunities.
In late December 2024, Tam Anh Research Institute launched the VISTA-1 project on oral immunotherapy for cancer treatment in Vietnam. After nearly six months of trial implementation, eight patients with end-stage colorectal cancer treated according to the research protocol have initially shown no significant side effects related to the research drug and have shown promising results.
Mr. M. (50 years old), who was the first person to test the drug in this project and suffered from colorectal cancer that had metastasized to the liver, said that he had undergone various treatment regimens such as chemotherapy and targeted therapy, but without success. In January 2025, he registered to participate in the study and was deemed eligible. "I thought I had no other options, but thanks to this opportunity to participate in the study, I feel relieved and have more hope," Mr. M. shared.
According to Dr. Vu Huu Khiem, Head of the Oncology Department at Tam Anh General Hospital in Hanoi , patients participating in the study were closely monitored for safety and response to the research drug. The research team has not recorded any unusual symptoms related to the research drug in patients who have used it for the past three months.
“For patients with advanced colorectal cancer who no longer respond to current treatment regimens, every improvement in survival time during their participation in this study is of great significance. We and the patients have now entered the fourth month of this research,” said Dr. Vu Huu Khiem.
At the Ho Chi Minh City Oncology Hospital, Dr. Phan Tan Thuan, Head of the Clinical Trial Unit, stated that the unit is currently conducting 37 clinical studies, mostly in collaboration with multinational pharmaceutical companies (Pharma). The hospital also proactively collaborates with regional cancer centers to conduct independent research with academic value and practical applications. The hospital often uses new generation drugs for comparison with standard treatment protocols. Many of these drugs are not yet available in Vietnam and are very expensive, with some costing up to 300 million VND per treatment cycle (3-4 weeks). Patients participating in the studies receive the medication free of charge, regardless of their specific treatment branch.
“Besides helping patients access modern treatment earlier, these studies also enable domestic professionals to master data and evaluate the effectiveness of drugs on the Vietnamese population. This shortens the time it takes to bring new drugs to market. Many new generation drugs are very expensive. Here, we provide free access to new generation drugs for all patients participating in the study. When the research is successful, it can shorten the time it takes to bring new drugs to market,” Dr. Phan Tan Thuan explained.
"Recent clinical trials have helped the healthcare industry rapidly access modern treatments, from traditional chemotherapy to immunotherapy and targeted therapies. These advances not only help prolong life but also improve the quality of life for patients, while expanding access to new drugs at lower costs for patients and the healthcare system."
Deputy Minister of Health Nguyen Thi Lien Huong
Much potential, but a lack of mechanisms.
According to Professor Guy Thwaites, Director of the Clinical Research Unit at Oxford University, Vietnam has been a leader in malaria drug trials since 1991, benefiting millions of patients globally. With a population of approximately 100 million, Vietnam provides a diverse patient group, ideal for clinical trials. An average GDP growth rate of 6-7% per year, coupled with a growing middle class, further enhances Vietnam's attractiveness to both domestic and international donors. Despite its great potential, the clinical trial market in Vietnam still faces several barriers that slow its development and create a lack of synchronization. The approval time for a clinical trial in Vietnam can be up to 160 days – the longest in Asia, while the average time in other countries is around 75 days. Furthermore, Vietnam lags far behind many countries in the region such as Singapore (18 days), Indonesia (20 days), Japan (31 days)... This timeframe affects the attractiveness of the Vietnamese market to research sponsors.
Furthermore, clinical trials require close collaboration between doctors, nurses, research coordinators, and data managers. However, Vietnam currently lacks a well-trained workforce; it also faces financial difficulties and inadequate incentive mechanisms. A phase 3 clinical trial can cost between $10 and $20 million. Meanwhile, many investors have reported that Vietnam lacks tax incentives, research funding, or co-financing mechanisms similar to those in Singapore and South Korea. Funding disbursement is often delayed due to administrative procedures, impacting trial progress and reducing competitiveness compared to other countries in the region.
To develop the clinical trial sector in Vietnam, Dr. Phan Tan Thuan suggests simplifying approval procedures through a centralized online system to shorten the current complex approval process. Additionally, online registration via an English-language portal and bilingual documentation should be implemented to streamline the process for international donors. To compete with other countries in the region, preferential policies such as tax exemptions, research funding, or co-funding mechanisms for trial costs, similar to programs in Singapore and Thailand, should be developed.
“Vietnam should promote public-private partnership models in clinical trial research to help share risks and enhance resources for sustainable industry development. Along with that, there is a need for policies to invest in infrastructure, create more testing facilities, and expand specialized training programs for personnel through cooperation with universities and international organizations,” suggested Dr. Phan Tan Thuan.
Mr. LUKE TRELOAR, Head of Infrastructure, Government and Healthcare at KPMG Vietnam: Developing incentive policies
The clinical trial approval process in Vietnam involves many steps. First, obtaining approval at the local level takes between 3 and 12 months, depending on the number and location of the study. Next, the approval process at the Ministry of Health takes an average of 3 to 5 months. Finally, obtaining an import license for the drug (for drug trials) takes an average of 3 to 4 months. The number of clinical trial centers and studies in Vietnam is too low relative to its population, risking falling behind in the race to become a regional clinical research center. Vietnam needs policies to invest in infrastructure, build more trial facilities, and expand specialized training programs for personnel through cooperation with universities and international organizations. To compete with other countries in the region, it needs to develop incentive policies such as tax exemptions, research funding, or co-financing mechanisms for trial costs, similar to programs in Singapore and Thailand.
Mr. Nguyen Ngo Quang, Director of the Department of Science, Technology and Training (Ministry of Health): Standardizing clinical trial activities.
In the coming period, the Ministry of Health will continue to refine the legal framework and standardize clinical trial activities in line with international standards. Simultaneously, it will strengthen the capacity of the National Biomedical Research Ethics Council. Currently, the Ministry of Health is seeking feedback on a draft Circular detailing regulations on clinical drug trials. This Circular will specify the details of clinical drug trials, including principles, standards, procedures, and responsibilities related to testing drugs on volunteers to assess the safety and efficacy of drugs, identify adverse drug reactions (ADRs), and study drug pharmacokinetics. According to the draft Circular, the principles of clinical drug trials must ensure scientific rigor, objectivity, honesty, transparency, and protect the rights of research participants.
MINH NAM - MINH KHA NG recorded
Source: https://www.sggp.org.vn/gian-nan-thu-nghiem-lam-sang-post802539.html















































































































Comment (0)